Print version
Search Pub Med
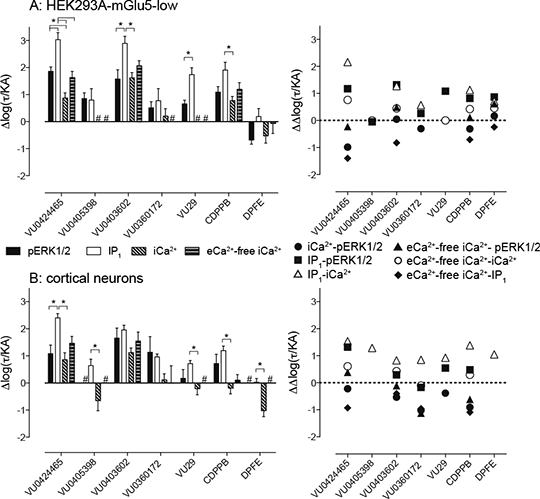
Biased allosteric agonism and modulation of metabotropic glutamate receptor 5: implications for optimising preclinical neuroscience drug discovery Emerging evidence for CNS disorders with glutamatergic dysfunction suggests the metabotropic glutamate receptor subtype 5 (mGlu5) is a promising target. Current mGlu5 allosteric modulators have largely been classified based solely on modulation of intracellular calcium (iCa2+) responses to orthosteric agonists, resulting in a narrow classification of ligands, and potential unappreciated bias in receptor signalling. We assessed seven diverse mGlu5 allosteric modulators across iCa2+, inositol phosphate (IP1) accumulation and phosphorylation of extracellular signal-regulated kinases (pERK1/2), to explore their potential for engendering biased agonism and/or modulation. Relative to the reference orthosteric agonist, (S)-3,5-dihydroxyphenylglycine (DHPG), all seven allosteric ligands exhibited biased agonism in HEK293A cells stably expressing mGlu5, coupling more strongly to IP1 accumulation and pERK1/2 over iCa2+ mobilisation. Disparities in iCa2+ and IP1 suggests the presence of ion channel modulation, rather than solely Gq activation. These bias profiles were generally recapitulated at native receptors in cortical neuron cultures. Relative to DHPG, (R)-5-((3-fluorophenyl)ethynyl)-N-(3-hydroxy-3-methylbutan-2-yl)picolinamide (VU0424465) showed significantly greater potency at IP1 accumulation (pEC50 = 8.70±0.13 vs 5.44±0.10, p<0.05 one-way ANOVA, Tukey's post-test, n=3-10), as well as pERK1/2 (pEC50 = 8.39±0.13 vs 6.31±0.25, p<0.05 one-way ANOVA, Tukey's post-test, n=3-10). Interestingly, N-(1,3-diphenyl-1H-pyrazolo-5-yl)-4-nitrobenzamide (VU29), and N-cyclobutyl-6-((3-fluorophenyl)ethynyl)picolin-amide (VU0360172), which showed no agonism for iCa2+ mobilisation in recombinant cells, showed robust agonism for both IP1 and pERK1/2 (logR=7.41±0.11, 6.44±0.39 (IP1) and 7.17±0.05, 7.02±0.18 (pERK1/2) respectively, n=3-10). Application of an operational model of agonism allowed quantification of biased agonism across mGlu5 signalling pathways relative to DHPG (Fig. 1). Unappreciated biased agonism and modulation may contribute to unanticipated effects (both therapeutic and adverse) when translating from recombinant systems to preclinical models.
Fig 1. mGlu5 allosteric ligands are biased agonists relative to DHPG in HEK293A-mGlu5-low and cortical neurons.
The transduction coefficient (log(τ/KA)) was derived as previously described (1) and normalized to that of DHPG (Δlog(τ/KA)). To calculate the degree of bias evident for different ligands between different pathways, Δlog(τ/KA) were subtracted from one another (ΔΔlog(τ/KA)) to determine Log bias factors. Data for Δlog(τ/KA) represent the mean ± SEM. Log bias estimates are mean only. * denotes significantly different comparisons, p<0.05, one-way ANOVA with Tukey's post-test. n=3-10 experiments performed in duplicate. (1) Kenakin T et al (2012). ACS Chem Neurosci 3(3): 193–203.
|