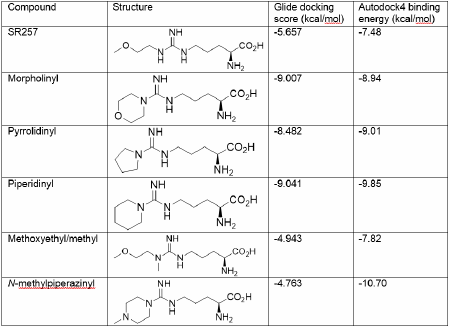
In silico and in vitro approaches to develop Dimethylarginine dimethylaminohydrolase-1 inhibitors Introduction: Dimethylarginine dimethylaminohydrolases (DDAH) metabolise the endogenous nitric oxide synthase (NOS) inhibitors: asymmetric dimethylarginine (ADMA) and monomethylarginine1. In sepsis excessive nitric oxide partially contributes to acute circulatory failure, and pharmacological DDAH1 inhibition has been proposed in order to increase methylarginines and reduce NO levels 2. The SR257 arginine analogue, with NG-methoxyethyl substituent, inhibits DDAH1 with an IC50 22 µM without directly inhibiting NOSs1,3. Method: Acyclic and cyclic NG,NG-disubstituted arginines were made as previously described4 using Katritzky’s synthesis preparing trisubstituted guanidines from di-(benzotriazol-1-yl)methanimine5. Molecular docking was employed to explore interactions of these NG,NG-disubstituted arginines with human DDAH1 (PDB 2JAJ) using Glide (Schroedinger6) and Autodock47. The published SR257 ligand was used to define the binding site with both software tools. Recombinant human DDAH1 activity was measured using colorometric citrulline assay8 containing ADMA (100 µM), sodium phosphate (10 mM pH7.4); with symmetric dimethylarginine (100 µM), not a substrate for DDAH1, as blank. Experiments were carried out in duplicate, and repeated on at least 3 separate occasions. Results: Recombinant DDAH1 activity was reduced to less than 25% of control (ADMA substrate, 100 µM) in the presence of 100 µM piperidinyl, methoxyethyl/methyl, N-methylpiperazinyl, with morpholinyl and pyrrolidinyl substituents reducing activity to less than 10% of control. The in silico Glide docking score and predicted Autodock4 binding energy for human DDAH1 (PDB, 2JAJ) for the known SR257 DDAH1 inhibitor and NG,NG-disubstituted arginines are shown in the table.
Conclusions: Both Autodock4 and Glide docking predicted higher binding energies for morpholinyl, pyrrolidinyl and piperinyl than the known SR257 compound. In vitro assays confirmed these NG,NG-disubstituted arginines reduced DDAH1 activity. There was variation between Glide and Autodock4 in the docking predictions for methoxyethyl/methyl and N-methylpiperazinyl. In silico prediction of DDAH1-ligand interactions may assist in the future design and development of novel NG,NG-disubstituted arginines. References: 1 Leiper, J. et al. (2007) Nat Med. 13:198-203. 2 Wang, Z et al. (2014) Biochem J. 460:309 3 Rossiter, S. et al. (2005) J Med Chem. 48:4670-4678. 4 Morfill, C et al. (2012) http://www.pA2online.org/abstracts/Vol10Issue4abst197P.pdf 5 Katritzky, A et al. (2000) J. Org. Chem. 65: 8080-8082. 6 Friesner, RA et al. (2006) J Med Chem. 49:6177-6196. 7 Morris, GM et al. (2009) J. Comp. Chem. 16:2785-91. 8 Knipp, M & Vasak, M (2000) Anal Biochem 286:257-64.
|